画像 chemistry homogeneous and heterogeneous mixtures 349774-Chemistry homogeneous and heterogeneous mixtures
Chemical solutions are usually homogeneous mixtures The exception would be solutions that contain another phase of matter For example, you can make a homogeneous solution of sugar and water, but if there are crystals in the solution, it becomes a heterogeneous mixture Many common chemicals are homogeneous mixturesActivity This one page worksheet will give your students practice with heterogeneous and homogeneous mixtures The topics of the questions include prefix meanings, examples, differences between the two types of mixtures, and the definition of mixtures The worksheet involves critical thinking skills and willMixtures are combination of two or more substances where each substance retains its individual physical properties There are two classes of mixtures homogeneous and heterogeneous Homogeneous mixtures have a constant composition whereas heterogeneous mixtures do not Another name for a homogeneous mixture is a solution
Mixture
Chemistry homogeneous and heterogeneous mixtures
Chemistry homogeneous and heterogeneous mixtures-Homogeneous Vs Heterogeneous Mixture In chemistry, we can have two types of mixtures homogeneous mixtures and heterogeneous mixtures Homogeneous mixture Blended so thoroughly, it looks like one substance – Uniform composition Heterogeneous mixture Not thoroughly blended, so you can see and pick out an individual part of the mixture A homogeneous mixture has the same uniform appearance and composition throughout Many homogeneous mixtures are commonly referred to as solutions A heterogeneous mixture consists of visibly different substances or phases The three phases or states of matter are gas, liquid, and solid An example of a Homogeneous mixture is brewed
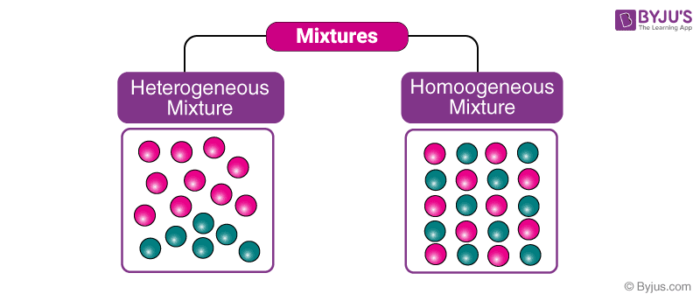



Heterogeneous And Homogeneous Mixture Differences Videos Examples
Q Define homogeneous mixture?A mixture is a combination of two or more substances in any proportions The substances in a mixture do not combine chemically, so they retain their physical properties A homogeneous mixture has the same composition throughout A heterogeneous mixture varies in its compositionThis chemistry video tutorial explains the difference between homogeneous and heterogeneous mixtures within the subtopic of the classification of matter It
Correct answer 2examples of homogeneous and heterogeneous mixtures eanswersincom Unlike homogeneous mixtures, heterogeneous mixtures do not have the same composition throughout Oil and water is a fine example of a heterogeneous mixture When oil is mixed with water, the separation of two layers is visible to the eyeAns A mixture which has uniform composition throughout its mass is called homogeneous mixture The word "homo" means "same" For example, when you completely dissolve salt in the water, you form a homogeneous mixture Q What is a heterogeneous mixture?
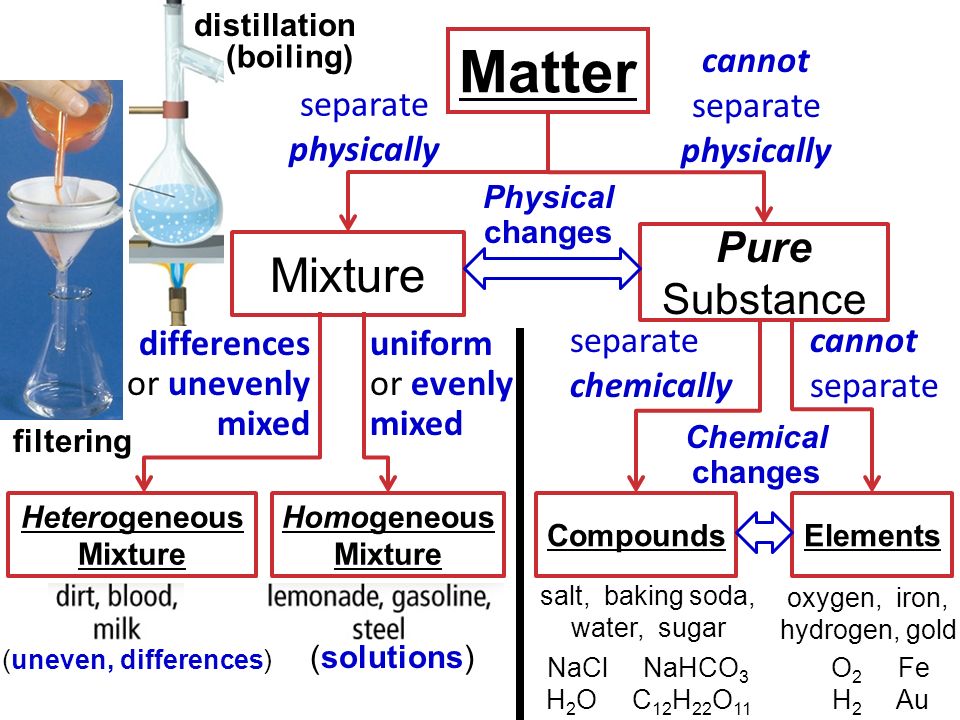
2) A homogeneous mixture does not contain physically distinct parts whereas a heterogeneous mixture contains physically distinct parts 3) Homogeneous mixtures form solutions whereas heterogeneous mixtures form colloids or suspensions 4) A mixture of alcohol and water is homogeneous whereas a mixture of sodium chloride and sand is heterogeneousClassify the following mixtures as homogeneous or heterogeneous a gasoline b a jar of jelly beans c chunky peanut butter d margarine e the paper on which this question is printed Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties Homogeneous mixtures are uniform, that is, their composition is the same wherever you look at it;




What Do You Need To Know About Heterogeneous And Homogeneous Mixtures




2 1 Homogeneous And Heterogeneous Mixtures
Homogeneous and Heterogeneous Mixtures Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world A mixture consists of sugar in water is a homogeneous mixture because we can't see particles of sugar in the water, as they are dissolved thoroughly A mixture consisting of oil in water is an example of the heterogeneous mixture as the oil cannot be mixed in the water and we can easily see them I hope you like my post about "Difference Homogeneous Mixtures are commonly known as solutions (since the solute and solvent are mixed thoroughly) On the other hand, heterogeneous mixtures are commonly known as suspensions and colloids In terms of physical properties, homogeneous mixtures possess the same nature Heterogeneous mixtures, on the other hand, don't showcase similar physical properties




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Difference Between Homogeneous Mixture And Heterogeneous Mixture
Homogenous vs Heterogeneous Matter created by Simmy8 on 21 Jun 12, enabled by jimmy Sciences Medium level (75% of success) 15 questions 52 037 players Classify the following substances and mixtures as either homogeneous or heterogeneousCheerios Trail mix Trail mix is HETEROGENEOUS Cheerios is homogeneous • Apple juice • Orange juice with pulp • Chocolate doughScience Chemistry Mixtures and Compounds Homogeneous and heterogeneous mixtures Create account Create a free account to watch the full video By signing up you will get access to this and many more videos and quizzes If you join a school with a membership you will also get access to every lesson



Sxxv4y Ycx9hem




Classification Of Matters With Examples Online Chemistry Tutorials
Homogeneous mixture Heterogeneous mixture 1) These are called as solutions These are called as suspensions/colloids 2) Substances are Uniformly distributed These substances are Unevenly distributed 3) These are not visible to the naked eye, but visible through the microscopeUniform appearance and composition throughout Many homogeneous mixtures are commonly referred to as solutions A heterogeneous mixture consists of visibly different substances or phases The three phases or states of matter are gas, liquid, The components of a homogenous mixture cannot be separated by simple physical means like filtration and sieving The components of a heterogeneous mixture can be separated by simple physical means like filtration Phases All the components of a homogenous mixture




Topic Chemistry Aim Describe Solubility And The Different Factors That Affect It Do Now Homogeneous Or Heterogeneous Mixture 1 Made Up Of Large Particles Ppt Download




Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Sorting Cards
Saltwater contains dissolved salt, so it's a homogeneous mixture as we can't separate salt from it directly But it is also called a heterogeneous mixture because of the presence of impurities and insoluble components like sands, shells made up ofQ Define a heterogeneous mixture? The way to know if a mixture is heterogeneous is by observing whether it has two or more material components or phases Examples of homogeneous mixtures are a plate of rice with lentils, cereals with milk, coca cola with ice, a mixture of




Heterogeneous Mixture High Res Stock Images Shutterstock



Heterogeneous Mixture Ppt Video Online Download
In chemistry, when two or more substances mix with each other without participating in a chemical change, the resulting substance is called a Mixture Mixtures can be classified into the following categories Homogeneous mixtures – possess the same properties and combination throughout their mass Heterogeneous mixtures – possess different properties and compositions in variousA homogeneous mixture is a mixture in which the composition is uniform throughout the mixture All solutions would be considered homogeneous A heterogeneous mixture is a mixture in which the composition is not uniform throughout thePlay this game to review Chemistry A combination of 2 or more substances that cannot be easily separated is called Mixtures Homogeneous and Heterogeneous DRAFT 8th grade 110 times Chemistry 69% average accuracy a year ago jameshorvath 0 Save Edit Edit Mixtures Homogeneous and Heterogeneous DRAFT a year ago by jameshorvath
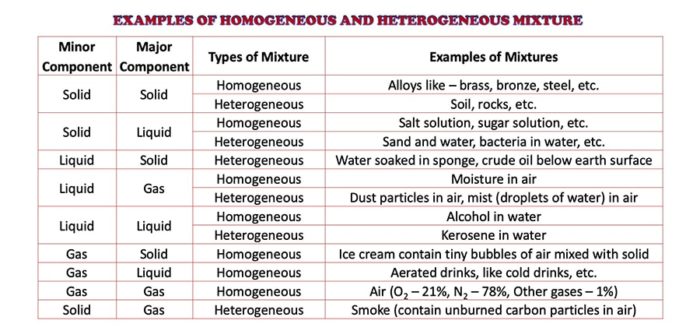



Homogeneous Heterogeneous Mixture Definition Examples Selftution




Homogeneous Mixture Experiment Qs Study
Homogeneous mixture Heterogeneous mixture It has a uniform composition It has a nonuniform composition It has only one phase There are two or more phases It can't be separated out physically It can be separated out physically 'homo' means the same 'hetero' means different Example a mixture of alcohol and waterIn many cases, the mixed elements can be easily separated again For example, a salad Unlike homogeneous mixtures, in heterogeneous mixtures it is very easy to identify, even with the naked eye, what are the different components that make them up This makes it much easier to separate these mixes at the same timeA chemical mixture combines two substances that maintain their own properties when combined Heterogeneous mixtures are made up of a nonuniform composition, while homogeneous mixtures are made up of a uniform compositionFor example, water and sand is a heterogeneous mixture — you can easily separate the sand from the water



Homogeneous And Heterogeneous Mixtures




Homogeneous Or Heterogeneous Worksheet
DRAFT 2 years ago by ledlightsStart studying Chemistry Mixtures Homogeneous/Heterogeneous Learn vocabulary, terms, and more with flashcards, games, and other study toolsHomogeneous chemistry basically refers to the homogeneous reactions that take place in various conditions So in order to understand the chemistry of homogeneous reactions, it is important to have a clear knowledge of homogeneous mixtures and solutions A homogeneous mixture can be defined as a mixture that has a uniform composition throughout




Types Of Mixtures In Terms Of Homogeneity Science Online
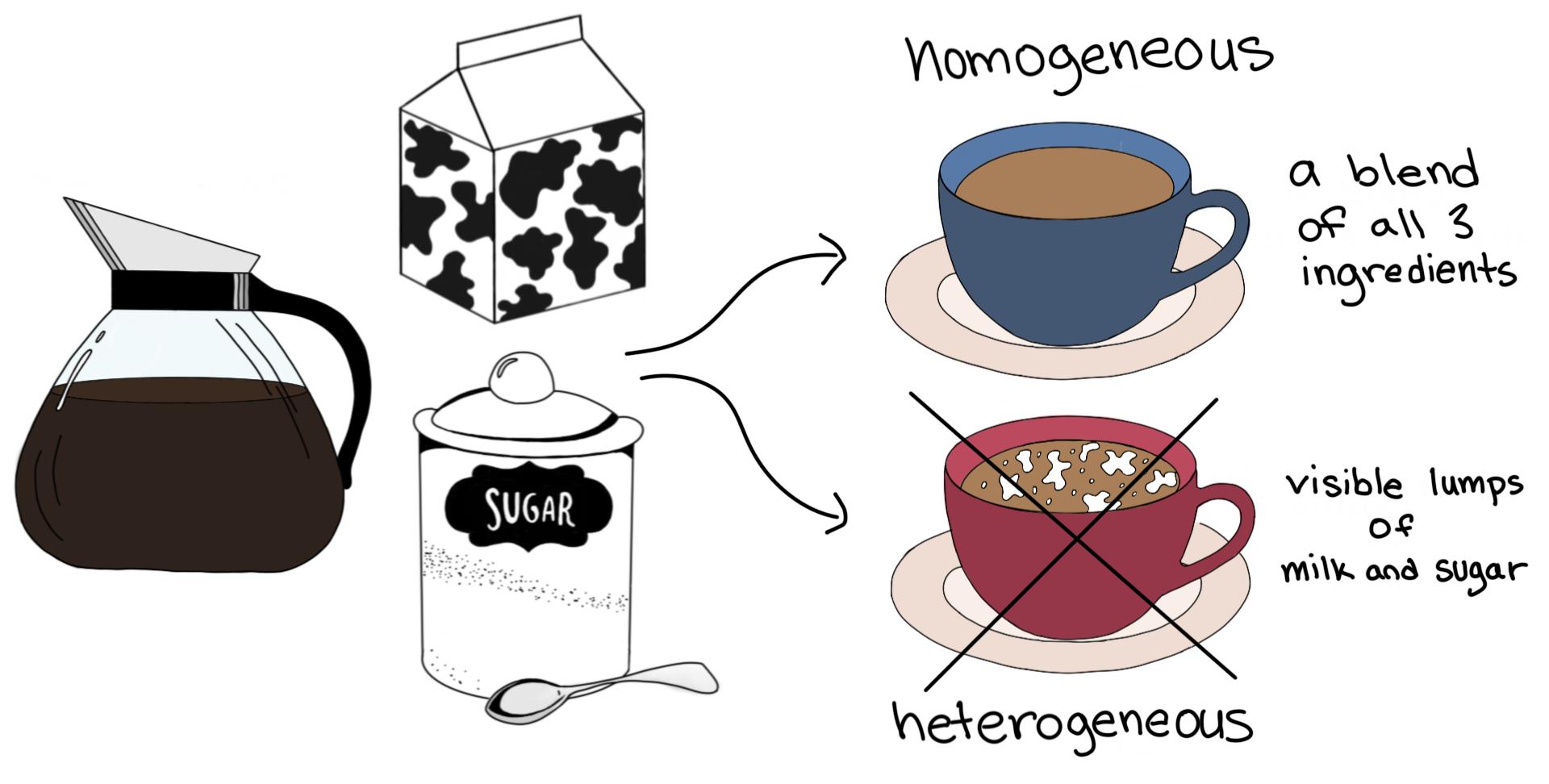



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
DRAFT 6th 8th grade Played 16 times 79% average accuracy Chemistry, Science 2 years ago by ledlights 9 Save Edit Edit Homogeneous or Heterogeneous Mixture?Play this game to review Chemistry Freshly brewed black coffee Preview this quiz on Quizizz Quiz Homogeneous or Heterogeneous Mixture?Homogeneous mixtures have uniform composition Heterogeneous mixtures have nonuniform composition What type of mixtures are these?
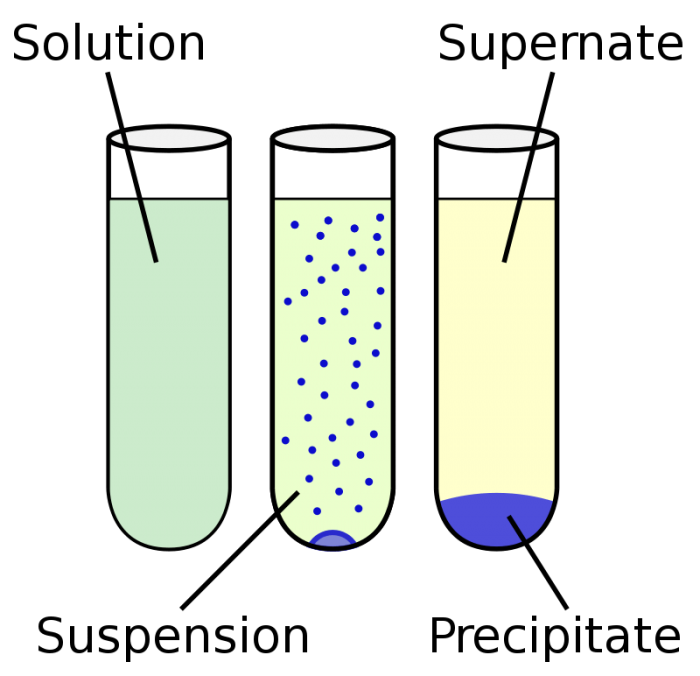



5 Examples Of Heterogeneous Mixtures For Chemistry Class Science Trends




Grade 6 Q1 Homogeneous And Heterogeneous Mixtures Homogeneity And Heterogeneity Mixture
In chemistry, we categorize all material around us as – pure substances and mixture Mixtures are further classified as – homogeneous mixture and heterogeneous mixture In this post, we will learn about the characteristics of the homogeneous and heterogeneous mixture along with examples The difference between heterogeneous and homogeneous mixtures is the degree to which the materials are mixed together and the uniformity of their composition A homogeneous mixture is a mixture in which the components that make up the mixture are uniformly distributed throughout the mixture The composition of the mixture is the same throughoutHomogeneous and heterogeneous mixtures In chemistry, if the volume of a homogeneous suspension is divided in half, the same amount of material is suspended in both halves of the substance An example of a homogeneous mixture is air In physical chemistry and materials science this refers to substances and mixtures which are in a single phase




Shutterstock Puzzlepix




Chemistry Heterogeneous Homogeneous Mixtures Glasses Salty Stock Vector Royalty Free
Within the categories of homogeneous and heterogeneous mixtures there are more specific types of mixtures including solutions, alloys, suspensions, and colloids Solutions (homogeneous) A solution is a mixture where one of the substances dissolves in the other The substance that dissolves is called the soluteA chemistry experiment demonstrates how to tell the difference between heterogeneous mixtures and homogeneous mixtures using a flashlight In chemistry, the difference between a homogeneous and heterogeneous mixture is a bit more complicated A homogeneous mixture has a uniform composition with all components in a single phase, while a heterogeneous mixture has a nonuniform composition with components in at least two different phases Properties of Heterogeneous Mixtures
.png?revision=1)



1 3 Classification Of Matter Chemistry Libretexts



Elements Compounds And Mixtures Course Hero
A heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture Vegetable soup is a heterogeneous mixture Vegetable soup is a heterogeneous mixture Any given spoonful of soup will contain varying amounts of the different vegetables and other components of the soup The mixture is homogeneous as long as there is only one phase (you see only liquid which looks like water but is really a solution of sugar in water) If you continue to add sugar, the extra sugar will not dissolve, but will fall to the bottom of the container Then, you will have a twophase system, which is heterogeneous Heterogeneous mixtures are mixtures with inconsistent, nonuniform composition The parts of a heterogeneous composition can be mechanically separated from each other Examples include salad, trail mix and granite A solution is when a homogeneous mixture of one or more substances (the solutes) dissolved in another substance (the solvent)




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube




Mr Chapman Chemistry 30 What Is A Solution A Solution Is A Mixture Or A Combination Of Two Or More Things A Solution Is Also Known As A Homogeneous Ppt Download
0128 Give the classification of matter when two substances exist with two phases present and the substance cannot be decomposed by chemical means heterogeneous mixture The density of table sugar is 159 g/mL It is true that 500 g of table sugar occupies a volume of 314 mL Sugar is a pure substance Because, it is composed of molecules of only one compound, i e, sugar (C12H22O11) So, it is not at all a mixture, it is a pure substance Every pure substance is homogeneous Because, composition and property of different portions of the substance are same through out the substance Hope, this helpsHeterogeneous mixture the substances are not distributed evenly (chocolate cups, pizzas, rocks) Within the categories of homogeneous and heterogeneous mixtures there are more specific types of mixtures including solutions, alloys, suspensions and colloids




Chemistry




Homogeneous High Res Stock Images Shutterstock
While heterogeneous mixtures are uneven, with a composition that varies from one point to another In homogeneous mixtures, there seems to be only one component (solute and solvent), but in heterogeneous, we easily visualize more than two components A heterogeneous mixture is one in which the composition of the mixture is not uniform all the way through Suspensions, colloids, and emulsions are types of heterogeneous mixtures When investigating these types of mixtures, students learn how to differentiate between a suspension and a colloid and what makes a colloid different from an emulsionThe difference between homogeneous mixtures and heterogeneous mixtures is a matter of scale the heterogeneous mixture can be seen on beaches where sand included many particles like coral, shells and organic matter, etc they all can be separated easily hence known as a heterogeneous mixture but when we take a large amount of sand, it's impossible to separate all the matter,



3




Homogeneous Mixture And Heterogeneous Mixture Is Matter Around Us Pure Chemistry Class 9 Youtube




Heterogeneous And Homogeneous Mixtures In Cooking And Learning Communities By Natalie King And Brandon Connelly Re Writing Chemistry




Post Lab Exercises 1 Classify As Element Compound Chegg Com
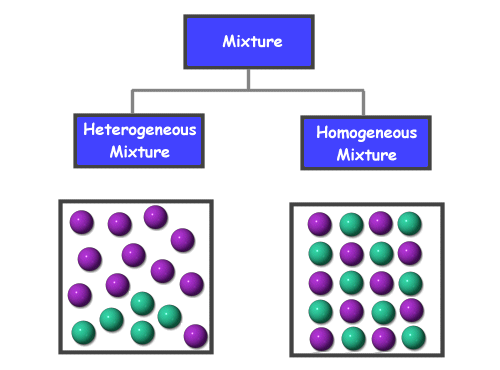



Chemistry For Kids Chemical Mixtures




Homogeneous And Heterogeneous Mixtures Activities




Part C Properties Of Homogeneous And Heterogeneous Chegg Com




What Is The Difference Between Heterogeneous Mixture Vs Homogenous Mixture Brainly Com
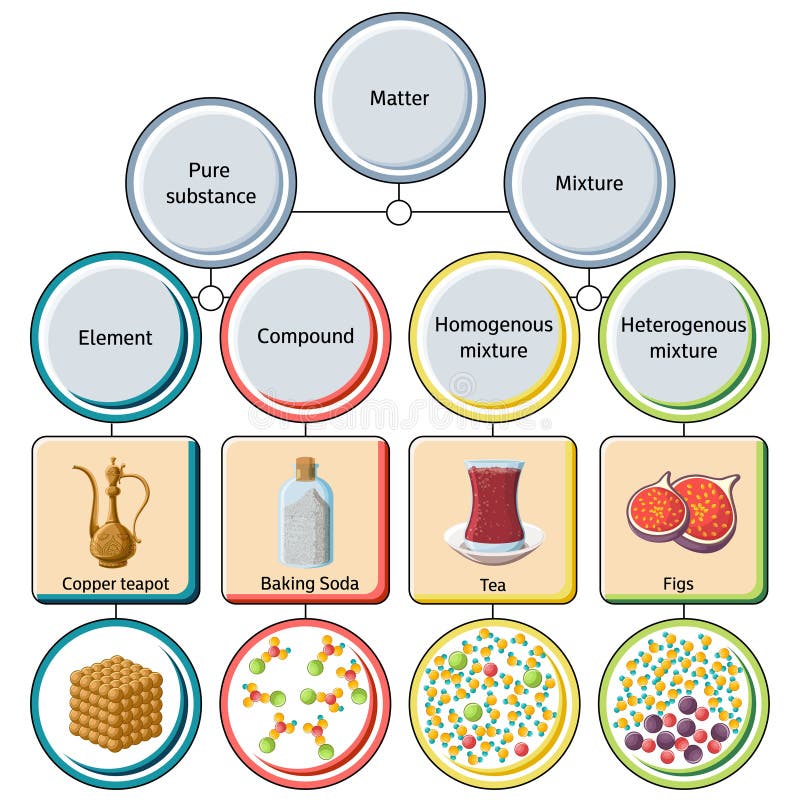



Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime




List The Points Of Difference Between Homogeneous And Heterogeneous Mixtures Brainly In
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples




Mixtures Worksheet Chemistry




Homogeneous Mixture Definition Examples Tutors Com




Homogeneous And Heterogeneous Mixtures Slides By The Weekly Sprinkle
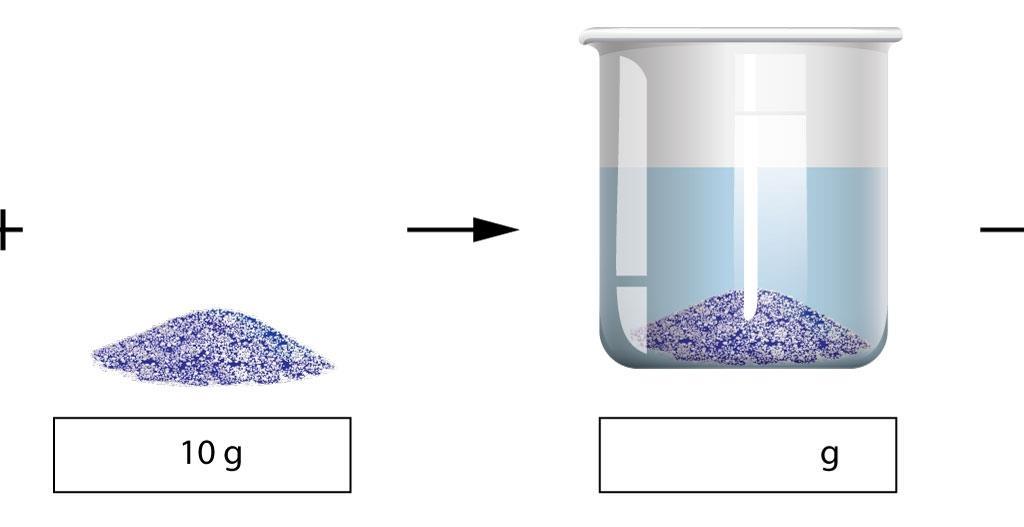



Mixtures And Solutions Cpd Rsc Education




Difference Between Homogeneous And Heterogeneous Compare The Difference Between Similar Terms
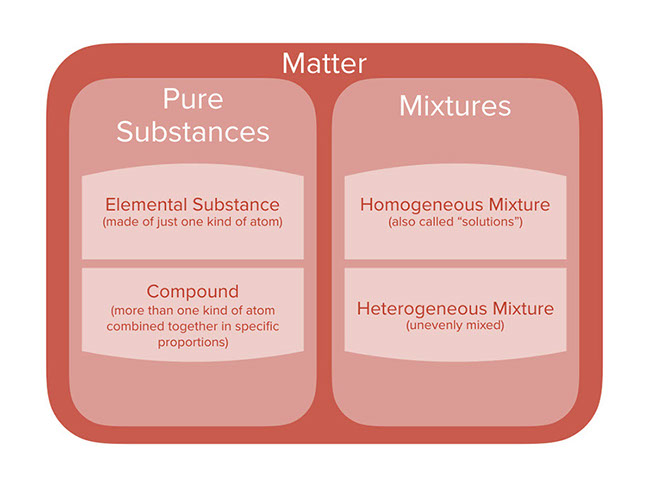



Lesson Categories Of Chemicals And Mixtures



Homogeneous And Heterogeneous Mixtures Properties Of Matter




Heterogeneous Mixture High Res Stock Images Shutterstock



Mixture



Is Sugar A Homogeneous Or Heterogeneous Mixture Chemistry Point
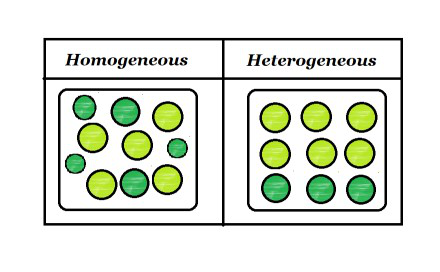



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




Ethiopia Learning Chemistry Grade 12 Page 7 In English



Homogeneous And Heterogeneous Mixture Nine Science




Elements Mixtures And Compounds Lab



Mixture




Examples Of Heterogeneous Mixtures Types Made Simple




Homogeneous Mixture Definition Lesson For Kids Video Lesson Transcript Study Com




Heterogeneous Homogeneous Mixture Card Sort For Matter In Chemistry




Homogeneous And Heterogeneous Mixtures Iken Edu Iken Iken App Youtube



3
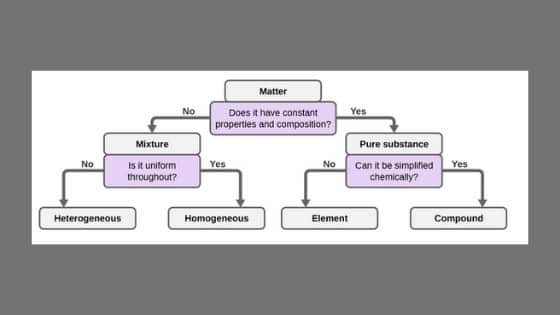



Homogeneous Mixture And Heterogeneous Mixture Ncert Books




This Presentation Discusses Homogeneous And Heterogeneous Mixtures Provides Examples Explains How A Particle Di Heterogeneous Mixture Pure Products Mixtures




Homogeneous Or Heterogeneous Mixtures Practice Worksheet




Examples Of Homogeneous Mixtures And Heterogeneous Mixtures Youtube



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




Chemistry Homogeneous And Heterogeneous Mixtures Handwiki



Difference Between Homogeneous And Heterogeneous Welding



Homogeneous Heterogeneous Mixture Definition Examples Selftution




Pure Substances And Mixtures 1 Pure And Mixed




10 Examples Of Mixtures
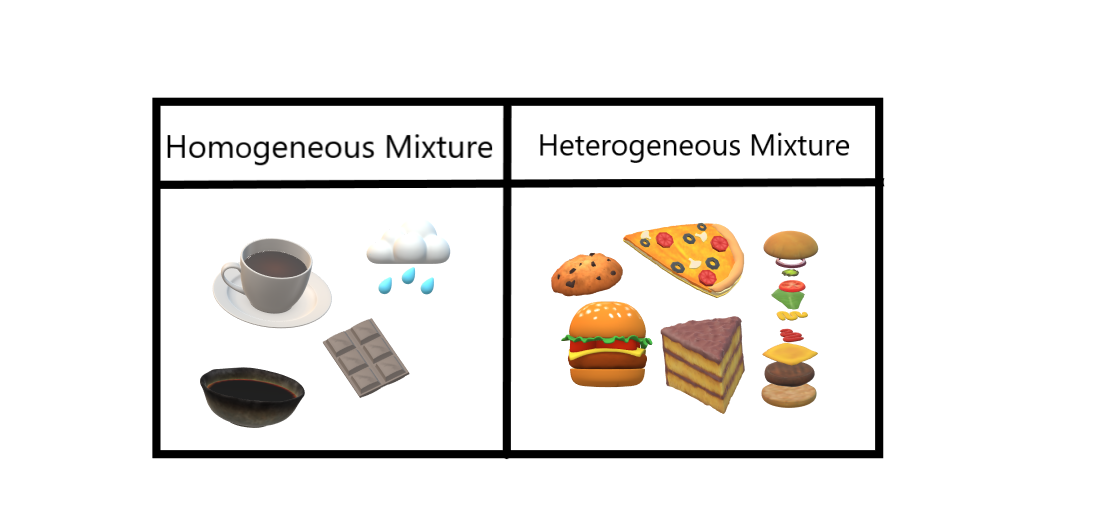



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




Classify Mixtures As Homogeneous Or Heterogeneous Worksheet




Difference Between Homogeneous And Heterogeneous Mixture Snapsolve



Q Tbn And9gctjuuyeomalljsdlz1u 5uxw8ihdx0o5wibwkoouvfqqtwvrcm3 Usqp Cau



Chemistry Lower Secondary Ydp Whiteboard Exercise Homogeneous And Heterogeneous Mixtures
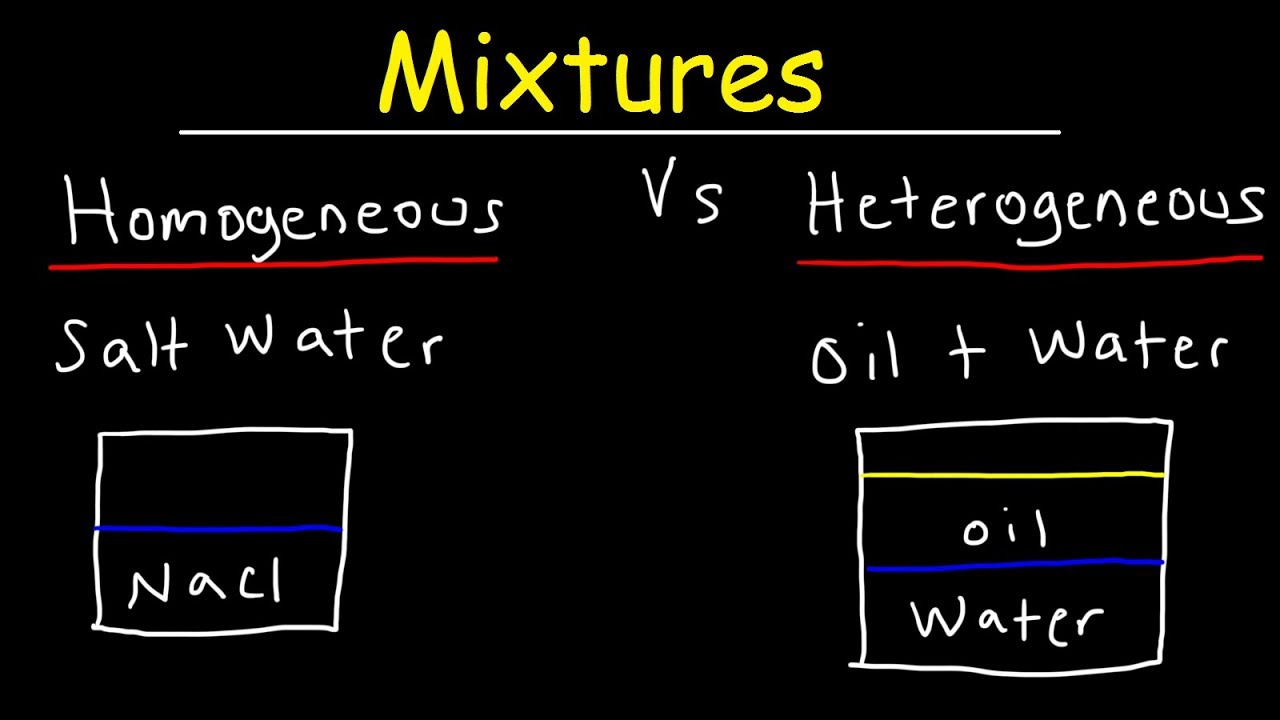



Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube



Heterogeneous Mixtures




List The Points Of Differences Between Homogeneous And Heterogeneous Mixtures
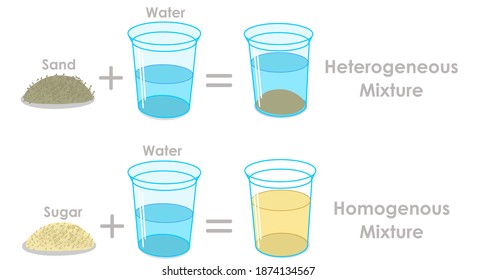



Mixtures Images Stock Photos Vectors Shutterstock




Homogenous Compounds And Mixtures Homogeneous Mixture Heterogeneous Mixture




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Homogeneous Vs Heterogeneous Mixture




Heterogeneous Mixture Homogeneous Mixture Worksheet Easy Hard Science




Homogeneous Mixture Definition Examples Tutors Com




Homogeneous And Heterogeneous Mixtures Card Sorting Activity By Elly Thorsen




Heterogeneous And Homogeneous Mixture Differences Videos Examples




Ways To Separate Mixtures Definition Types Homogeneous Heterogeneous Mixture Eschool
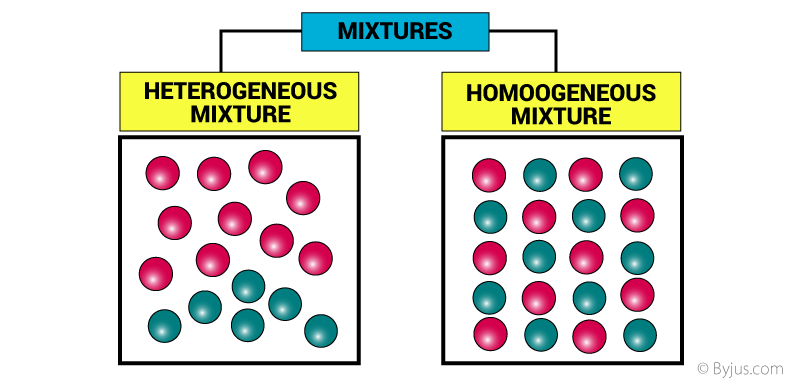



What Is A Mixture Definition Properties Examples Types With Videos




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
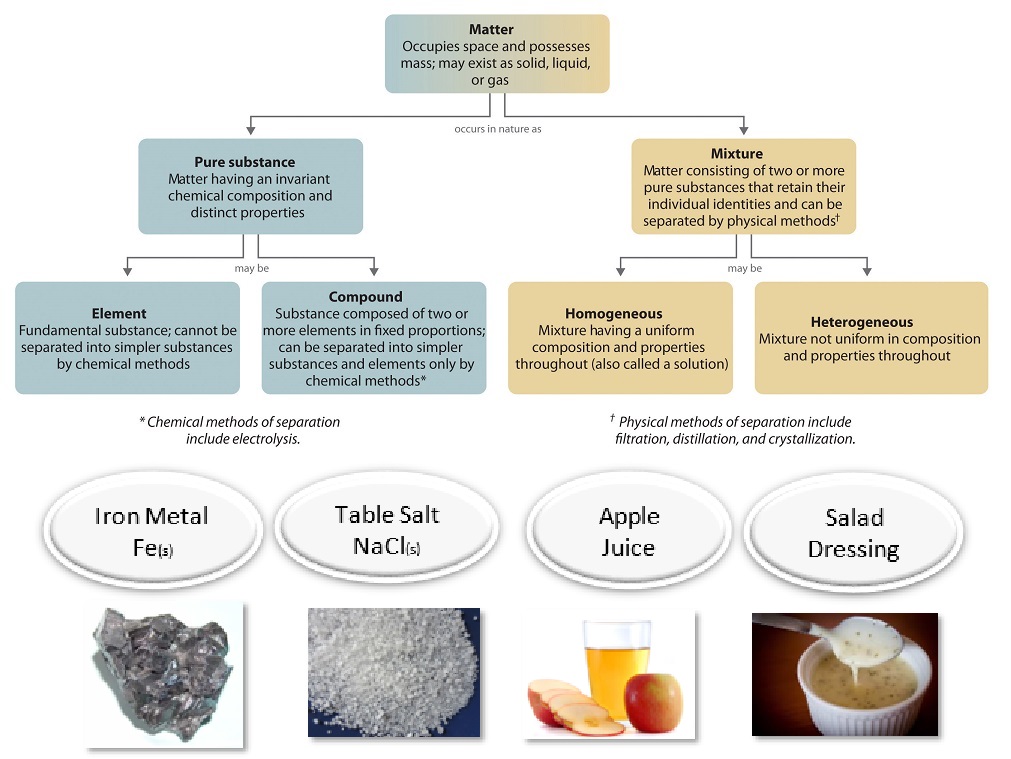



3 4 Classifying Matter According To Its Composition Chemistry Libretexts



Mixture




Homogeneous And Heterogeneous Mixtures Properties Of Matter




3 5 Pure Substances And Mixtures Chemistry Libretexts



1




Compound Vs Mixture Difference And Comparison Diffen



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




Homogeneous And Heterogeneous Mixtures Png Images Pngwing




What Is A Homogeneous Mixture Definition And Examples




Classification Of Matter Chemistrygod




Pure Substances Elements Compounds Homogenous Heterogenous Mixture Examples And Problems Youtube




What Is A Heterogeneous Mixture Definition And Examples
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures



Simple English Chemistry Basics




Chemistry Chapter 1 Flashcards Quizlet




Heterogeneous Mixture And Homogeneous Mixture Youtube




Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube




Heterogeneous Homogeneous Mixtures
コメント
コメントを投稿